USA Biological Indicator Vials Demand to Reach USD 91.4 Million by 2035 | Regional Demand & Forecast Scenarios
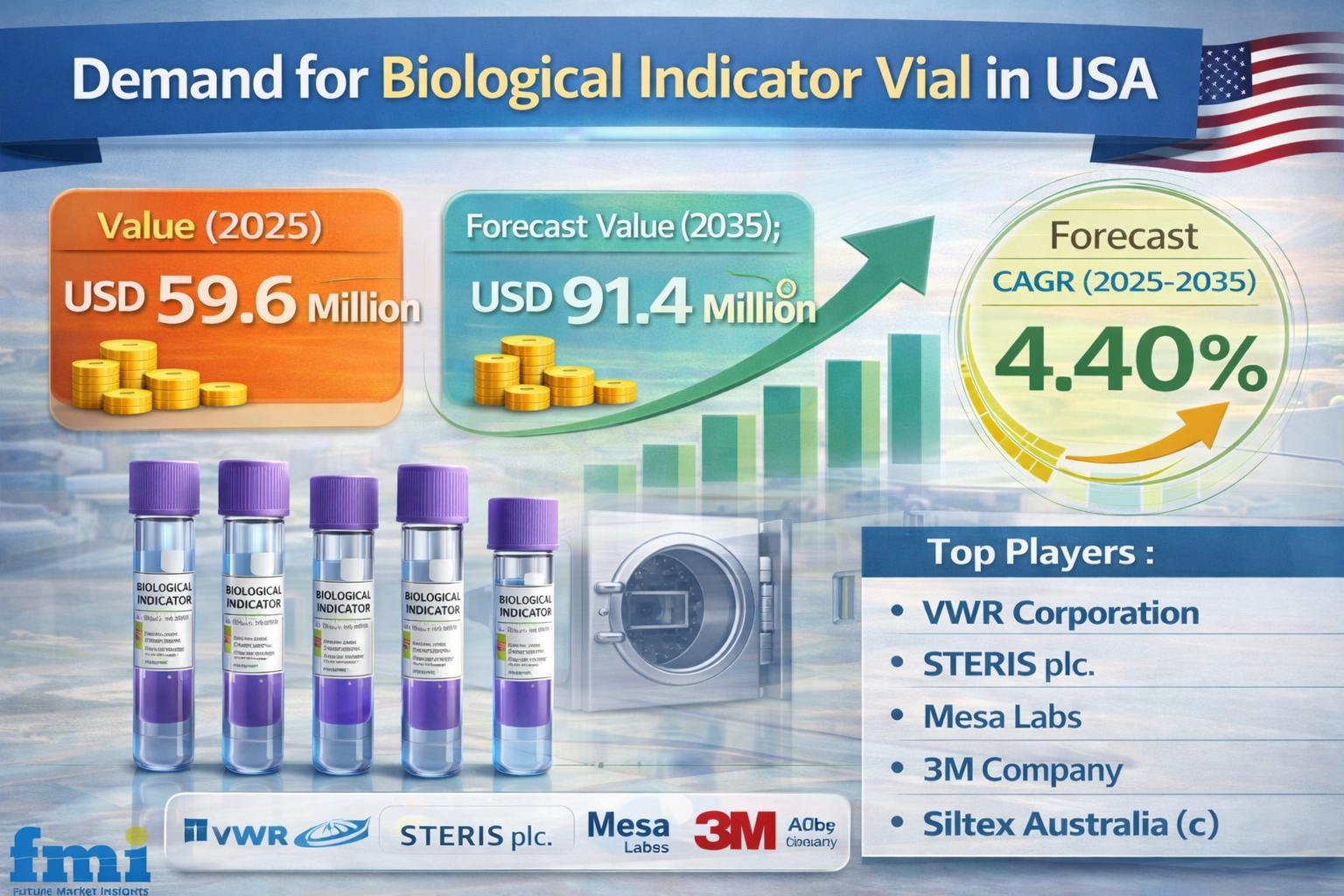
The Demand for Biological Indicator Vials in the USA is projected to grow from USD 59.6 million in 2025 to USD 91.4 million by 2035, reflecting a steady CAGR of 4.4%. Growth is being driven by stricter sterilization validation requirements across pharmaceuticals, biotechnology, healthcare, and medical device manufacturing, where sterility is essential for regulatory compliance and patient safety. Biological indicator vials serve as critical validation tools in high-risk environments such as hospitals, laboratories, and sterile manufacturing facilities, ensuring sterilization processes effectively eliminate microbial contamination.
- Market size in 2025? USD 59.6 million
- Market size in 2035? USD 91.4 million
- CAGR (2025–2035)? 4.40%
- Leading sterilization type? Steam sterilization (57%)
- Leading incubation time? 24 to 32 hours (45%)
- Leading end use? Pharmaceutical industry (55%)
- Key growth regions? West, South, Northeast, Midwest
- Top players? VWR Corporation; STERIS plc.; Mesa Labs; 3M Company; Siltex Australia (c)
Market Momentum (YoY Path)
The Demand for Biological Indicator Vials in the USA follows a two-phase growth pattern. From USD 59.6 million in 2025, demand increases steadily to USD 62.2 million by 2030, adding USD 2.6 million over the first five-year block. Between 2030 and 2035, demand accelerates significantly, rising from USD 62.2 million to USD 91.4 million, contributing an additional USD 29.2 million. This acceleration is driven by stronger regulatory enforcement, rising sterilization procedures, and advanced sterilization technologies in pharmaceutical and healthcare sectors.
Why the Demand for Biological Indicator Vials in the USA is Growing
Growth is rooted in increasing sterilization validation requirements across healthcare and regulated manufacturing. Hospitals, pharmaceutical companies, and laboratories rely on biological indicator vials to verify the effectiveness of sterilization cycles for surgical instruments, injectable drugs, and sterile medical devices. Rising surgical volumes, expansion in biopharmaceutical manufacturing, and stricter accreditation standards are intensifying reliance on validated sterilization monitoring. Technological advancements such as rapid-read indicators and enhanced spore systems are also supporting workflow optimization and faster compliance verification.
Request For Sample Report | Customize Report | Purchase Full Report - https://www.futuremarketinsights.com/reports/sample/rep-gb-28687
Segment Spotlight
Steam Sterilization Leads with 57%: Steam sterilization accounts for 57% of total demand. Its effectiveness, safety, and consistent sterilization performance make it the preferred method in pharmaceutical and healthcare environments. Biological indicator vials are essential in confirming the elimination of resistant microbial spores during steam cycles, reinforcing compliance and quality assurance.
24 to 32 Hours Incubation Dominates with 45%: The 24 to 32 hours incubation time segment holds 45% share. This duration balances microbial detection accuracy with operational efficiency, making it ideal for high-throughput sterilization environments. Faster turnaround supports improved workflow management in pharmaceutical and healthcare facilities.
Pharmaceutical Industry Leads with 55%: The pharmaceutical sector represents 55% of demand. Sterility validation is critical for vaccines, injectable drugs, and medical products. As regulatory standards intensify and sterile manufacturing expands, biological indicator vials remain central to quality assurance processes.
Drivers, Opportunities, Trends, Challenges
- Drivers: Increasing sterilization procedures, regulatory scrutiny, and growth in sterile manufacturing (including biologics and advanced therapies) are propelling demand. Healthcare-associated infection prevention initiatives further strengthen adoption.
- Opportunities: Emerging manufacturing segments such as cell and gene therapy require validated sterilization workflows, increasing demand for indicator vials. Rapid-read technologies and data integration capabilities broaden application scope.
- Trends: Technological innovations focus on faster incubation times, higher sensitivity spore systems, and improved traceability through barcoding and laboratory information system integration. Automation in monitoring systems is gaining traction.
- Challenges: Premium rapid-read vials carry higher costs, limiting adoption in smaller facilities. Validation complexity and documentation requirements increase operational burden. Supply-chain constraints for specialized consumables may also moderate growth.
Competitive Landscape
The competitive environment in the Demand for Biological Indicator Vials in the USA is shaped by sterilization validation expertise, regulatory compliance capabilities, and product reliability. VWR Corporation holds approximately 30.3% share, supported by its distribution strength and consumables portfolio. Other key contributors include STERIS plc., Mesa Labs, 3M Company, and Siltex Australia (c). Companies compete on performance reliability, rapid-read innovation, and sterilization-type compatibility.
Market participants focus on expanding sterilization verification technologies, improving spore-based systems, and enhancing digital traceability to align with modern quality management frameworks.
Scope of the Report
- Quantitative Unit: USD million
- Sterilization Type: Steam Sterilization; Ethylene Oxide Sterilization
- Incubation Time: 24 to 32 Hours; Up to 24 Hours; 32 to 48 Hours; Above 48 Hours
- End Use: Pharmaceutical; Food & Beverages; Cosmetics
- Regions Covered: West; South; Northeast; Midwest
- Key Companies Profiled: VWR Corporation; STERIS plc.; Mesa Labs; 3M Company; Siltex Australia (c)
- Additional Attributes: Dollar sales by sterilization type, incubation time, end use, and regional trends
Why FMI: https://www.futuremarketinsights.com/why-fmi
Have a Look at Related Research Reports on the Packaging Domain:
USA Labels Market: https://www.futuremarketinsights.com/reports/united-states-labels-market-share-analysis
Demand for Box Liners in USA: https://www.futuremarketinsights.com/reports/united-states-box-liners-market
USA Stick Packaging Market: https://www.futuremarketinsights.com/reports/united-states-stick-packaging-market
Contact Us:
Future Market Insights Inc.
Christiana Corporate, 200 Continental Drive,
Suite 401, Newark, Delaware - 19713, USA
T: +1-347-918-3531
For Sales Enquiries: sales@futuremarketinsights.com
Website: https://www.futuremarketinsights.com
About Future Market Insights (FMI)
Future Market Insights, Inc. (FMI) is an ESOMAR-certified, ISO 9001:2015 market research and consulting organization, trusted by Fortune 500 clients and global enterprises. With operations in the U.S., UK, India, and Dubai, FMI provides data-backed insights and strategic intelligence across 30+ industries and 1200 markets worldwide.
- Arte
- Causas
- Artesanía
- Bailar
- Bebidas
- Película
- Fitness
- Alimento
- Juegos
- Jardinería
- Salud
- Hogar
- Literatura
- Musica
- Redes
- Otro
- Fiesta
- Religión
- Compras
- Deportes
- Teatro
- Bienestar



