-
Feed de notícias
- EXPLORAR
-
Páginas
-
Grupos
-
Eventos
-
Reels
-
Blogs
-
Marketplace
-
Jobs
Therapeutic Drug Monitoring Market to Expand by 47.4% Growth, Reaching USD 2.86 Billion by 2035
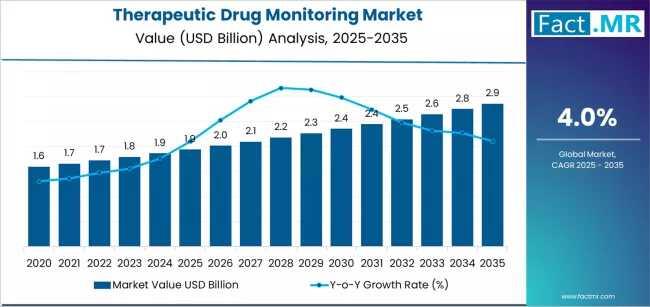
The global therapeutic drug monitoring (TDM) market is undergoing a precision-led transformation, projected to grow from USD 1.94 billion in 2025 to USD 2.86 billion by 2035. According to a comprehensive analysis by Fact.MR, the market is set to expand by nearly 1.5X over the next decade, driven by a paradigm shift toward personalized treatment protocols and an uncompromising focus on patient safety optimization.
As healthcare providers move away from "one-size-fits-all" dosing, TDM has evolved into a critical clinical pillar. By measuring drug concentrations in the blood, clinicians can now optimize dosing in real-time, significantly reducing adverse drug reactions (ADRs) and improving therapeutic efficacy for drugs with narrow therapeutic windows.
Get Access Report Sample :
https://www.factmr.com/connectus/sample?flag=S&rep_id=11757
Quick Stats: Therapeutic Drug Monitoring Market at a Glance
- Market Value (2025E): USD 1.94 Billion
- Projected Value (2035F): USD 2.86 Billion
- Forecast CAGR:0% (2025–2035)
- Leading Product Segment: Consumables (65.4% share)
- Dominant End-User: Hospitals (60.0% share)
- Growth Hotspot: India (6.0% CAGR)
Product Insights: Consumables and Immunoassay Dominance
The market’s revenue structure is heavily weighted toward recurring needs, with consumables forming the bedrock of daily clinical operations.
Consumables (65.4% share):
This segment leads due to the continuous demand for reagents, kits, and assays required for routine monitoring. Healthcare providers prioritize consumables that offer high analytical reliability and operational efficiency.
Equipment (34.6% share):
While consumables drive volume, equipment drives the technological frontier. Immunoassay analyzers represent 50% of the equipment breakdown, favored for their rapid testing capabilities and seamless integration into automated hospital workflows.
Therapeutic Spotlight: Antiarrhythmic Drugs Take the Lead
Different drug classes require varying levels of scrutiny. Cardiovascular therapies currently necessitate the highest degree of monitoring precision.
- Antiarrhythmic Drugs (45.0% share): This is the primary drug class driving adoption. Given their narrow therapeutic windows and inter-patient variability, precise monitoring is essential to prevent toxicity while ensuring cardiac stability.
- Immunosuppressants (30.0% share): A vital segment in transplant medicine, where maintaining specific drug levels is the difference between organ rejection and long-term graft survival.
- Antiepileptic Drugs: Continues to be a staple of the TDM market as clinicians manage chronic seizure disorders with complex pharmacokinetic profiles.
Regional Performance: Asia-Pacific Outpacing Global Averages
While the USA (3.8% CAGR) remains a mature market leader in personalized medicine adoption, India and China are emerging as the world's most dynamic growth engines.
|
Country |
Projected CAGR |
Primary Growth Catalyst |
|
India |
6.0% |
Rapid infrastructure development and government-backed quality initiatives. |
|
China |
5.5% |
Aging population demographics and massive hospital diagnostic upgrades. |
|
South Africa |
4.5% |
Increasing investment in chronic disease management and infectious disease monitoring. |
|
Brazil |
4.2% |
Expanding diagnostic center networks and rising chronic disease prevalence. |
|
Germany |
3.5% |
Advanced healthcare systems and strong regulatory support for precision dosing. |
Market Dynamics: Drivers and Strategic Challenges
Key Drivers:
- Personalized Medicine: The move toward "Precision Dosing" is making TDM a standard requirement for complex treatment regimens.
- Patient Safety: Global initiatives to reduce medication errors and toxicity are mandating more rigorous monitoring protocols.
- Automation: The integration of automated laboratory systems and AI-driven clinical decision support is improving turnaround times.
Market Restraints:
- Capital Costs: High initial investments for sophisticated analytical equipment can challenge smaller diagnostic facilities.
- Technical Complexity: The need for specialized training to interpret complex pharmacokinetic data.
- Standardization Issues: Variability in monitoring protocols across different clinical settings.
Competitive Landscape: The "Big Three" and Beyond
The competitive field is led by Abbott (15.0% share), followed by global heavyweights like Thermo Fisher Scientific, F. Hoffmann-La Roche Ltd, and Siemens Healthineers. Competition is shifting from hardware specs to "integrated clinical utility," where manufacturers provide not just the test, but the data integration tools necessary for immediate clinical action.
To View Related Report:
Demand for Therapeutic Drug Monitoring in UK https://www.factmr.com/report/united-kingdom-therapeutic-drug-monitoring-market
Therapeutic Support Surface Market https://www.factmr.com/report/therapeutic-support-surface-market
Therapeutic Catheters Market https://www.factmr.com/report/2690/therapeutic-catheters-market
Gout Therapeutics Market https://www.factmr.com/report/gout-therapeutics-market
- Contact Us -
11140 Rockville Pike, Suite 400, Rockville,
MD 20852, United States
Tel: +1 (628) 251-1583 | sales@factmr.com
About Fact.MR
Fact.MR is a global market research and consulting firm, trusted by Fortune 500 companies and emerging businesses for reliable insights and strategic intelligence. With a presence across the U.S., UK, India, and Dubai, we deliver data-driven research and tailored consulting solutions across 30+ industries and 1,000+ markets. Backed by deep expertise and advanced analytics, Fact.MR helps organizations uncover opportunities, reduce risks, and make informed decisions for sustainable growth.
- Arte
- Causas
- Artesanía
- Bailar
- Bebidas
- Película
- Fitness
- Alimento
- Juegos
- Jardinería
- Salud
- Hogar
- Literatura
- Musica
- Redes
- Otro
- Fiesta
- Religión
- Compras
- Deportes
- Teatro
- Bienestar



